|
Back to Blog
Clf3 molecular geometry5/28/2023  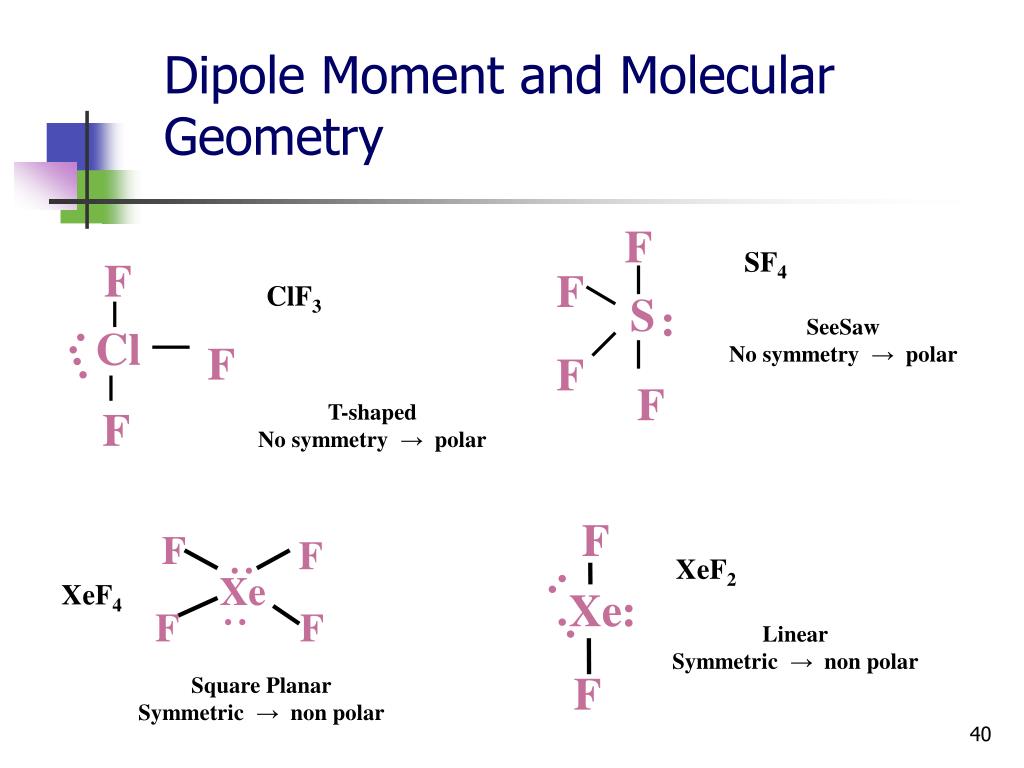
Child and Adolescent Psychology (PSY-355).Intro to Statistical Analysis (MAT-133).Creating and Managing Engaging Learning Environments (ELM-250).Intro to Biomedical Statistics (BST 322).Foundational Literacy Skills and Phonics (ELM-305).Introduction to Structured Query Language (DAD220).(v) Argon is used to provide inert atmosphere, helium is used in filling balloons for meteorological observations, neon is used in discharge tubes and fluorescent bulbs. The best-characterised compounds are those of xenon with fluorine and oxygen only under certain conditions. (iv) Due to complete octet of outermost shell, group 18 elements have less tendency to form compounds. (iii) All the noble gases except Rn occur in atmosphere. (ii) Electronic configuration: n s 2 n p 6 except He which has configuration of 1 s 2. (i) Elements: He, Ne, Ar, Kr, Xe, Rn In some cases X = O bonds are also found.Ĥ. Group 18 elements (Nobles gases or inert gases) In the structures of these oxoacids, halogen is the central atom which is bonded in each case with one OH bond as X – OH. (vii) A number of oxoacids of halogens are formed. (vi) H C l is prepared by heating N a C l with concentrated H 2 S O 4. (v) Chlorine is obtained by the reaction of H C l with K M n O 4. (iv) Halogens form oxides, hydrogen halides, interhalogen compounds and oxoacids. However, highest oxidation state can be + 7. (iii) The common oxidation state of these elements is – 1. It is used in the manufacture of several compounds. Amongst them, the most important is H 2 S O 4. It is used in the manufacture of H 2 S O 4. S O 2 is prepared by the reaction of sulphur with oxygen. (v) Sulphur combines with oxygen to give oxides such as S O 2 and S O 3. Allotropic form of oxygen is O 3 which is a highly oxidising agent. (iv) Dioxygen is prepared by heating K C l O 3 in presence of M n O 2. (ii) Electronic configuration: n s 2 n p 4 The oxoacids which have P–H bonds are good reducing agents.Ģ. Group 16 elements (Oxygen Family - Chalcogens) Depending upon the number of P–OH groups, their basicity varies. (xi) Phosphorus forms a number of oxoacids. (x) P C l 3 is prepared by the reaction of white phosphorus with dry chlorine and P C l 5 is prepared by the reaction of phosphorus with S O 2 C l 2. It forms two types of halides as P X 3 and P X 5. It forms hydride, P H 3 which is a highly poisonous gas. (ix) Phosphorus exists as P 4 in elemental form. Metals and non-metals react with H N O 3 under different conditions to give NO or N O 2 (viii) HNO 3 is a strong monobasic acid and is a powerful oxidising agent. (vii) Ammonia can be prepared on large scale by Haber’s process. These oxides have resonating structures and have multiple bonds. (vi) Nitrogen forms oxides in various oxidation states: N 2 O, N O, N 2 O 3, N O 2, N 2 O 4 and N 2 O 5. (v) Nitrogen differs from other elements of group 15 due to small size, formation of pπ – pπ multiple bonds with itself and with highly electronegative atom like O or C and non-availability of d orbitals to expand its valence shell. + 3 oxidation state is favoured by heavier elements due to ‘ inert pair effect’. (ii) Electronic configuration: n s 2 n p 3 The p-block elements consist of elements of groups 13 − 18 having n s 2 n p 1 - 6 as valence shell electronic configuration where n = 2 − 6. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
